Acne exacerbation after tofacitinib treatment for alopecia areata
HTML: 15
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
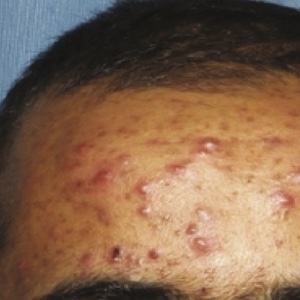
The major discovery of the novel therapeutic Janus kinase (JAK) inhibitors has been implicated in several dermatological diseases. Recently, studies have shown the efficacy and encouraging results of oral JAK inhibitors as a treatment for alopecia areata (AA). Due to the novelty of this treatment, potential side effects are not fully explored. In this paper, we present a case of a 28-year-old male with a 10-year history of alopecia totalis (AT) treated successfully with tofacitinib with encouraging effects on hair regrowth; however, a significant worsening of the patient’s facial acne was observed four months after AT treatment initiation. JAK inhibitors have promising results in the management of different dermatological conditions including moderatesevere forms of AA with few reported adverse events. Acne exacerbation is a unique observed adverse effect of this therapy. More thorough larger sized studies are needed to further characterize the association between acne exacerbation and the use of JAK inhibitors.
Solimani F, Meier K, Ghoreschi K. Emerging Topical and Systemic JAK Inhibitors in Dermatology. Front Immunol 2019;10:2847. DOI: https://doi.org/10.3389/fimmu.2019.02847
Damsky W, King BA. JAK inhibitors in dermatology: The promise of a new drug class. J Am Acad Dermatol 2017;76:736-44. DOI: https://doi.org/10.1016/j.jaad.2016.12.005
Samadi A, Ahmad Nasrollahi S, Hashemi A, et al. Janus kinase (JAK) inhibitors for the treatment of skin and hair disorders: a review of literature. J Dermatol Treat 2017;28:476‐83. DOI: https://doi.org/10.1080/09546634.2016.1277179
Cinats A, Heck E, Robertson L. Janus Kinase Inhibitors: A Review of Their Emerging Applications in Dermatology. Skin Therapy Lett 2018;23:5-9.
Mahajan S, Hogan JK, Shlyakhter D, et al. VX‐509 (decernotinib) is a potent and selective Janus kinase 3 inhibitor that attenuates inflammation in animal models of autoimmune disease. J Pharmacol Exp Ther 201;353:405‐14. DOI: https://doi.org/10.1124/jpet.114.221176
Shreberk-Hassidim R, Ramot Y, Zlotogorski A. Janus kinase inhibitors in dermatology: A systematic review. J Am Acad Dermatol 2017;76:745-53. DOI: https://doi.org/10.1016/j.jaad.2016.12.004
Ramot Y, Zlotogorski A. [JAK inhibitors for the treatment of alopecia areata]. Harefuah 2020;159:38-42.
Kontzias A, Laurence A, Gadina M, O'Shea JJ. Kinase inhibitors in the treatment of immune-mediated disease. F1000 Med Rep 2012;4:5. DOI: https://doi.org/10.3410/M4-5
Jabbari A, Dai Z, Xing L, et al. Reversal of alopecia areata following treatment with the JAK1/2 inhibitor baricitinib. EBioMedicine 2015;2:351–5. DOI: https://doi.org/10.1016/j.ebiom.2015.02.015
Liu LY, Craiglow BG, Dai F, King BA. Tofacitinib for the treatment of severe alopecia areata and variants: a study of 90 patients. J Am Acad Dermatol 2017;76:22–8. DOI: https://doi.org/10.1016/j.jaad.2016.09.007
Almutairi N, Nour TM, Hussain NH. Janus Kinase Inhibitors for the Treatment of Severe Alopecia Areata: An Open-Label Comparative Study. Dermatology 2019;235:130-6. DOI: https://doi.org/10.1159/000494613
Guo L, Feng S, Sun B, et al. Benefit and risk profile of tofacitinib for the treatment of alopecia areata: a systemic review and meta-analysis. J Eur Acad Dermatol Venereol 2020;34:192-201. DOI: https://doi.org/10.1111/jdv.15937
Tsai HR, Lu JW, Chen LY, Chen TL. Application of Janus Kinase Inhibitors in Atopic Dermatitis: An Updated Systematic Review and Meta-Analysis of Clinical Trials. J Pers Med 2021;11:279. DOI: https://doi.org/10.3390/jpm11040279
Rothstein B, Joshipura D, Saraiya A, et al. Treatment of vitiligo with the topical Janus kinase inhibitor ruxolitinib. J Am Acad Dermatol 2017;76:1054-60. DOI: https://doi.org/10.1016/j.jaad.2017.02.049
Awad SM, Tawfik YM, El-Mokhtar MA, et al. Activation of Janus kinase signaling pathway in acne lesions. Dermatol Ther 2021;34:e14563. DOI: https://doi.org/10.1111/dth.14563
PAGEPress has chosen to apply the Creative Commons Attribution NonCommercial 4.0 International License (CC BY-NC 4.0) to all manuscripts to be published.





 https://doi.org/10.4081/dr.2022.9396
https://doi.org/10.4081/dr.2022.9396



